Facts:
- Funding: Novo Nordisk Foundation
- Funding period: 2021-2024
Genes are transferred from parent to offspring, but genetic material can also be transferred between organisms in other ways, for example through bacteriophages (viruses that infects bacteria) and plasmids (DNA molecules within a cell separated from chromosomal DNA). This so-called horizontal gene transfer is the primary mechanism for the spread of antibiotic resistance in bacteria, and improved knowledge of the mechanisms involved could open for novel biotech and biomedical applications.
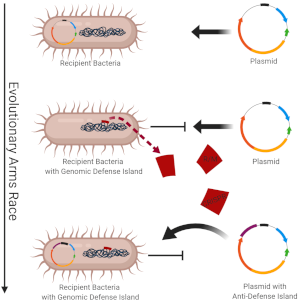
Horizontal gene transfer (HGT) of mobile genetic elements such as conjugative plasmids shapes bacterial evolution and adaptation, and therefore has a tremendous impact on both biotechnical and healthcare sectors. As plasmids can carry genes both beneficial and detrimental to their bacterial host, most prokaryotes possess defence mechanisms against invading DNA acting as barriers to HGT. However, plasmids have evolved anti-barriers that overcome the microbial defence systems.
A better understanding of this evolutionary arms race could help us explain the fate of plasmids in microbial communities and improve our understanding of phenomena such as antimicrobial resistance gene dissemination.
The research project PEACE will:
The results of PEACE will provide fundamental knowledge instrumental for biotechnical applications such as targeted microbiome engineering or gene editing. Discovery of new molecular defence and anti-defence systems will most likely also open for novel biotech and biomedical application of these systems.
The project is led by Professor Sören Sörensen at the University of Copenhagen.