Contact
Mikaela Gönczi, Director
SLU Centre for Pesticides in the Environment
mikaela.gonczi@slu.se, +46 18-67 31 05
What happens to pesticides (plant protection products) that are sprayed on arable land? The pesticides remain in the environment for a shorter or longer period and in some cases can even make their way far from the place where they were used.
Pesticides are often found in rivers or streams but also in the groundwater. Residues from pesticides have even been found in rain. The risk of unwanted spread of pesticides to the environment is mainly related to cropping and spraying techniques and also to natural conditions and processes (climate, soil properties, etc.).
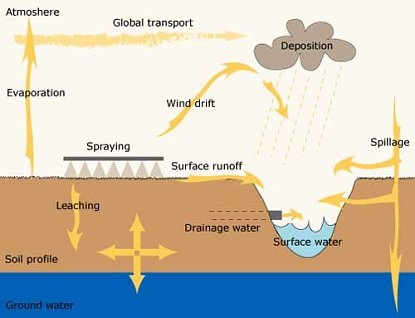
Figure. Pesticide distribution pathways in the countryside (Modified after Lennart Torstensson, SLU)
Some of the pesticides sprayed on fields can be transported to the surrounding land and water. Pesticides can be transported away in the air through wind drift and evaporation and then carried back to soil or water by rainfall. Surface run-off and percolation of water to the drainage system can carry pesticides out into rivers and lakes. Leaching of water through the soil can carry pesticides down to the groundwater. Various spillages during handling can also lead to pesticides going astray into the environment. Figure: Modified after Lennart Torstensson, SLU
Pesticides can be transported away in the air through wind drift and evaporation and then carried back to soil or water by rainfall.
Some of the pesticides sprayed on a field are blown away by the wind. Measurements have shown that between 1 and 5% of the amount sprayed is usually lost through wind drift. The single most important factor is wind speed at the time of spraying. An increase in wind speed of a few metres per second can double the amount of pesticide carried away with the wind. There is then a risk of the pesticide causing direct damage to the surrounding flora and fauna or being spread farther in the environment.
A proportion of the pesticide can evaporate from soil and crop to the air. Measurements have shown that up to 10% of the amount sprayed is usually lost through wind drift.

Figure. The atmosphere contains traces of pesticides.
Swedish studies have shown that around 0.01-1% of the total amount of pesticides used in an area can be spread to neighbouring streams via surface run-off and leaching.
Pesticides can accompany water running off the surface of the soil to the nearest drainage channel or water course. During heavy rain, the soil can become saturated and rainwater then runs off the surface, carrying with it soluble substances and soil particles (erosion). The longer the time between spraying and heavy rain, the lower the risk of the pesticide ending up in streams and rivers. In addition, the risk of surface run-off is related to the soil type in the sprayed field. Structurally weak silty and fine sandy soils have a greater risk of surface run-off than clay soils with a stable structure or coarse sands with high permeability. Surface run-off can be of great local significance in transport of pesticides to nearby waters, but this process is probably limited to occasional high rainfall events and to sites where there is a particular risk of surface run-off. Surface run-off often occurs in connection with snow-melt when the ground is frozen. Persistent substances that were sprayed during the previous growing season can then be transported away via surface run-off. There are a range of countermeasures that can be effective to reduce losses, including grass-covered buffer zones. International studies show that local conditions in the form of topography, soil properties, tillage and crop are critical in determining the suitability of different countermeasures.
Any water that cannot be stored in the soil following rainfall or irrigation is transported downwards in the soil profile and ends up in drainage pipes or is carried onwards to the groundwater. Pesticides dissolved in the soil solution are carried with the moving water. The risk of drainage losses is greatest in soils where the water can percolate easily. These include e.g. sandy soils and fractured clay soils.

Figure. Through surface run-off and leaching to drainage pipes, pesticides can be carried out into streams and rivers. By leaching through the soil, they can also end up in groundwater.
Through wormholes, cracks and root channels, rainwater can be rapidly transported down into the soil profile to subsurface drains opening into adjoining ditches or rivers and to the groundwater. This phenomenon is known as macropore flow and means that any pesticides accompanying water in its downward movement can reach surface water and groundwater fairly quickly, without having been broken down chemically or biologically. The greatest risk of macropore flow leaching crack-filled exists in cracked, often clayey soils that can swell and shrink, though macropore flow leaching can occur in most soil types.

Figure. By dyeing water applied to the surface, it is possible to monitor water movements in the soil. The red streaks in the soil profile show the macropores in this soil. Photo: Nick Jarvis, SLU.
A number of different soil properties affect the breakdown and mobility of pesticides. Soil composition and properties vary depending on geological origins, position in the landscape, climate zone and the various cropping or soil management practices used. The risk of leaching increases considerably if, for example, binding is weak due to lack of organic matter in the soil or breakdown is slow due to the microbial activity being low. In soils with a high clay content and high organic matter content, the conditions are often better for rapid breakdown of pesticides. For acid or alkaline substances, soil acidity (pH) affects the chemical form of the pesticide and its water solubility and thereby its mobility. The pH of the soil can also affect the degradation of pesticides.
Soil properties also affect the length of time pesticides can remain in the soil without being broken down, i.e. their lifetime or persistence. The soil is a complex and variable environment which greatly affects the soil solution, dissolved compounds, gases and microorganisms. Mineral particles and organic material make up the solid phase of the soil, while soil water and soil air are found in the soil pores between the solid particles. The soil is continually being affected by temperature changes, weathering, living organisms, water percolation and substances being added from the atmosphere.
The length of time a pesticide remains in the soil without being broken down mainly depends on how accessible it is for breakdown by microorganisms. In the smallest soil cavities, those less than one-tenth of a micrometre, pesticides can survive for a long time. This is because soil microorganisms are simply too large to enter these small pores. The pesticide is therefore physically protected against breakdown. It is mainly clayey soils that have a large proportion of such small, inaccessible micro-cavities.
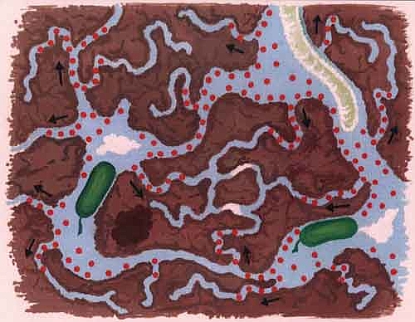
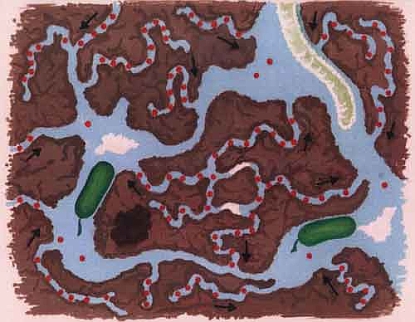
Figure. In pores so small that microorganisms (green in the pictures) cannot enter them, pesticides can survive for a long time. The top diagram shows pesticide distribution (red dots) after spraying. The bottom diagram shows pesticide distribution following microbial decomposition Pictures from: Bergström & Stenström, 1998. Environmental Fate of Chemicals in Soil. Ambio Vol. 27 No 1.
Pesticides are broken down in different ways and at different rates. The risk of accumulation and spread in the environment decreases the faster a pesticide is broken down. Breakdown of pesticides can occur in the plant, air, soil and water, mainly in one of three ways:
Breakdown occurs both within and outside organisms. Some microorganisms can adapt to using certain pesticides as an energy source for their own growth (metabolic breakdown). This has been demonstrated for example for the herbicide MCPA, which is a phenoxy acid. The herbicide is then broken down more quickly in the years after the first spraying, i.e. the compound is not as long-lasting. This adaptation in microorganisms can continue even after a field is no longer sprayed. For example, field trials have shown that adaptation to MCPA as an energy source is still detectable after five years.
Breakdown can also occur through microorganisms, plant roots etc. exuding breakdown enzymes (co-metabolic breakdown) to the surrounding soil. These enzymes are actually produced to utilise some other energy substrate but at the same time contribute to the breakdown of pesticides. This type of breakdown does not lead to specific adaptations in soil organisms and therefore breakdown does not occur more quickly after repeated applications of the pesticide. This is probably the most common type of decomposition, including in sediment. The rate of breakdown is related to the availability of energy substrate in the soil/sediment. General biological activity in a soil (production of carbon dioxide per day) is a rough measure of the ability of a soil to co-metabolically break down pesticides.
Certain microorganisms also affect the soil pH through their metabolism. For example, breakdown of organic nitrogen to nitrate leads to a pH decrease in the soil. Such pH changes in turn affect the persistence of pesticides, i.e. their lifetime in the soil.
The chemical compounds exuded by plant roots and microorganisms can also affect the breakdown of pesticides. For example, they may include signalling compounds that affect other organisms, inhibiting or stimulating their activity.
Pesticide properties affect the risk of unwanted spread in the environment. The volatility of substances (vapour pressure) determines how much of a pesticide will evaporate to the atmosphere during and after application. This in turn affects the risk of a compound spreading over a wide area. Water solubility and adsorption capacity (capacity to bind to soil particles, particularly humus and clay) greatly affect the risk of compounds ending up in surface water and groundwater. High water solubility increases the risk of the compounds accompanying the water on its path down to the groundwater and to subsurface drains. High adsorption capacity, on the other hand, means that the compounds are not carried away as easily by water, although in conditions causing erosion and surface run-off they can easily end up in water courses. The persistence of a compound (its endurance) determines how long a pesticide will remain in the environment without being broken down. Substances with a low adsorption capacity (poor binding to the soil) and those with slow rate of breakdown pose the greatest risk of spread.
When assessing the risk of a pesticide ending up in surface water or groundwater, risk assessment tools, including those developed by the SLU, are available.
Mikaela Gönczi, Director
SLU Centre for Pesticides in the Environment
mikaela.gonczi@slu.se, +46 18-67 31 05