Contact
Department of Molecular Sciences, Livsmedelsbioteknologi
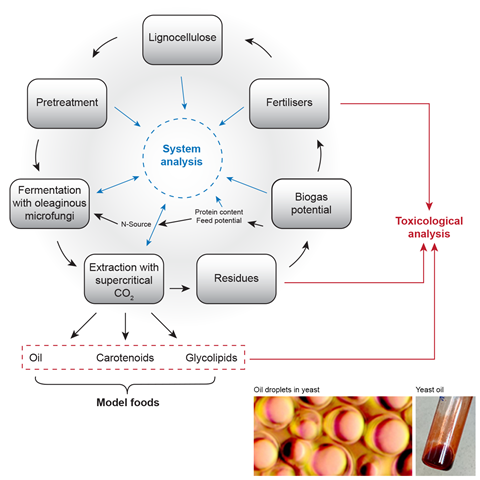
Production of future food and chemicals must rely on renewable resources, in a sustainable, circular process. Novel microbial cell factories can produce food, feed and biochemicals such as lipids and carotenoids from side products of agriculture, forestry and biofuel production, e.g. lignocellulose or crude glycerol. Microbial oils and protein can replace soya protein and –oil or palm oil. Food fermentation can provide plant- and fungal-based alternatives to meat. We also investigate safety aspects of these novel products.
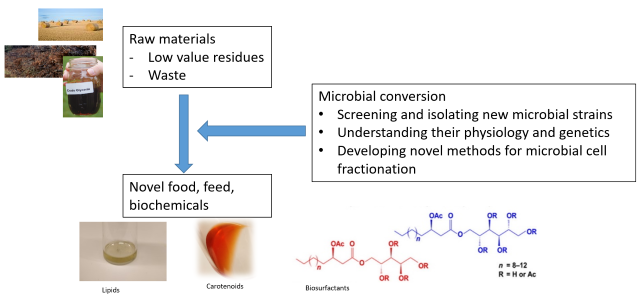
Our general goal is to use microbial diversity to establish sustainable, circular processes for the production of food, feed and biochemicals. This involves applied research to convert raw materials to valuable compounds and fundamental science to understand physiology and genetics of non- conventional microorganisms.

From left: PhD- students Mathilde Brunel, Yashaswini Nagaraj and Giselle Martín-Hernández, senior researchers Bettina Müller and Volkmar Passoth, and research engineers Nils Mikkelsen and Mikołaj Chmielarz.
Head of the unit
Volkmar Passoth, Professor
YouTube video: Food biotechnology - the origin and future of human civilisation
Senior researchers
Johanna Blomqvist, PhD
Su-Lin Hedén (Leong), Assoc. prof.
Bettina Müller, Assoc. prof.
Jana Pickova, Professor em.
PhD- students
Mathilde Brunel
Giselle Martín-Hernandez
Yashaswini Nagaraj
Research- engineers
Mikołaj Chmielarz, PhD
Nils Mikkelsen, PhD
Alumni
Jule Brandenburg, PhD (defence 2021)
Mikołaj Chmielarz, PhD (defence 2021)
Viktoriia Burkina, PhD
Fermentation
Microbial cultivation is the key for identifying microbes appropriate for biotechnological processes, understanding their physiology and establishing biotechnological processes. We have equipment for rapid screening microbes, and bioreactors for cultivating them under controlled conditions, from 200 ml scale up to 6 L working volume. This enables us to run physiological investigation and to provide microbial biomass for subsequent experiments, such as feeding trials.

Lipid lab and analytics
We have state of the art equipment for lipid analysis- FT-NIR spectroscopy for measuring intracellular lipid content, thin-layer chromatography (TLC) for determining lipid classes, UHPLC and spectrophotometer for measuring carotenoids and other substances.

Supercritical fluid (SFE) extraction unit
Supercritical fluid extraction can be used for extraction of hydrophobic compounds such as lipids or carotenoids from microbes and other biomaterial without using organic solvents.

Nanopore- sequencing
MinION is an Oxford Nanopore sequencing device, which uses a flow cell containing an array of nanopores embedded in an electro-resistant membrane. It is able to determine DNA or RNA sequences in short time, generating from small to ultra-long (>4 Mb) reads.
These long reads enable us to build chromosome-level genome assemblies of biotechnologically interesting strains and it can also aid in genetic engineering studies.

Extraction with supercritical CO2 enables effective separation of lipids and carotenoids without using organic solvents. We are establishing these methods as a basis of efficient cell fractionation as a step towards sustainable production of lipids, carotenoids and other high- value compounds from red oleaginous yeasts. Establishing these methods will also help to understand the physiology of lipid and carotenoid formation in those yeasts.

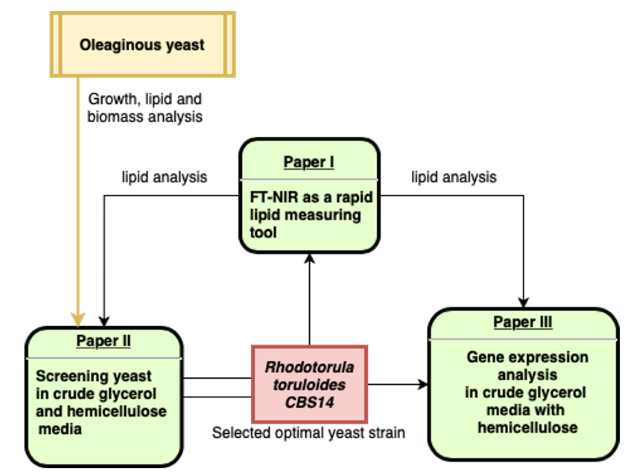
This PhD- project was about microbial lipid production from several lignocellulosic materials. Methods for fermentation and determination of intracellular lipid content were established. A variety of oleaginous yeast strains were tested on different lignocellulosic material and a substantial diversity of the lipid accumulation physiology was detected. The thesis was defended May 7, 2021 (https://publications.slu.se/?file=publ/show&id=111449)
The goals of this thesis were to establish a rapid, FT-NIR- based analysis tool for lipid quantification, investigate the conversion potential of crude glycerol (CG) and hemicellulose hydrolysate (HH) to lipids by oleaginous yeasts and to get an insight into physiology of conversion of CG and HH to yeast lipids. We found among others that addition of hemicellulose activates glycerol metabolism in Rhodotorula yeasts. The FT-NIR method can be used in basidiomycetous oleaginous yeasts such as Rhodotorula spec. as well as in ascomycetes such as Lipomyces starkeyi or Yarrowia lipolytica. The thesis was defended September 6, 2021 (https://pub.epsilon.slu.se/24912/)
The SAFE- project is a Nordic research project involving participants from Norway (NMBU, project coordination, Cargill, ForTek); Estonia (University of Tartu), Iceland (University of Akureyri) and Sweden (SLU).
It aims at developing and evaluating a novel multicomponent microbial biomass as a salmon feed from Nordic woody feedstock. SAFE will develop a process for producing multicomponent microbial biomass enriched with lipids, carotenoid pigments and β-glucans. Microbes investigated include oleaginous yeasts (production of oil, carotenoids and β-glucans) and Thraustochytrids (production of long-chain polyunsaturated fatty acids)
SLU project team: Johanna Blomqvist, Sabine Sampels, Volkmar Passoth
If you see mould on dairy cheese, it is okay to cut off the mouldy part plus 2 cm, and eat the rest. But is it safe to do the same with vegan cheeses? Note that vegan cheeses have coconut oil as a key ingredient, and coconut oil is known to support good mycotoxin production. This project is funded by Formas, and compares the ability for plant-based analogues (cheese, crème fraiche, paté) to support mould growth and mycotoxin production, compared with the original products. The extent of mycotoxin migration into the matrix is also quantified.
Project members:
Su-Lin Hedén, Johanna Blomqvist (SLU)
Åsa Svanström, Roland Lindqvist (Swedish Food Agency)
Michael Sulyok (IFA-TULLIN, BOKU Vienna)
Principal supervisor Jana Pickova
Projects on aquafeed development to improve sustainability
- Cultured oleaginous yeast as lipid source to fish feed
Taste of fish species from different rearing systems
- “off-flavour” analyses
Cytochrome P450 enzymes and their regulation by different natural and chemical compounds as an experimental tool
Fatty acid pathways in experimental trials
- Fish

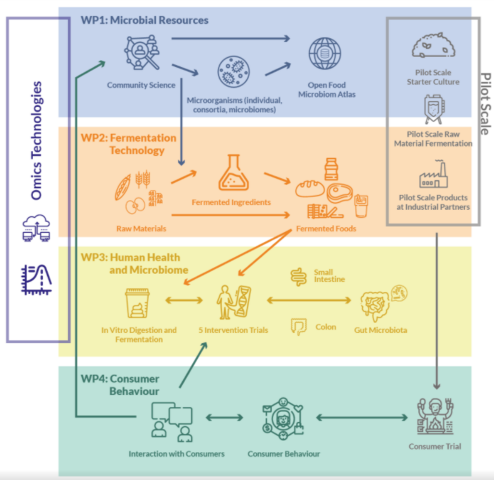
HealthFerm is a project within the Horizon FARM2FORK program of the EU. It involves partners from Belgium, Danmark, Finland, France, Germany, The Netherlands, Italy, Romania, Sweden, and Switzerland.
The overall objective of Healthferm is to enable an evidence-based transition in society and industry from traditional to sustainable plant-based fermented foods and diets that deliver health benefits to consumers by design.
In the frame of the project, we will test the ability of several microorganism to improve the nutritional value of Faba beans and oat and to develop fermentation processes for obtaining a tempeh-like product.
SLU-project team: Maud Langton (coordinator), Galia Zamaratskaia, Volkmar Passoth

LivsID is an industry PhD-program, dedicated to research and education within food-related topics. It was initiated by the Swedish Government as a part of the National Food strategy. The program involves nine companies and seven departments at SLU. The Food Biotechnology Unit is coordinating the program, project team Volkmar Passoth, Bettina Müller, Johanna Blomqvist, Eva-Marie Hemming (administration).
Brunel M, Burkina V, Pickova J, Sampels S, Moazzami A (2022) Oleaginous yeast Rhototorula toruloides biomass effect on the metabolism of Arctic char (Salvelinus alpinus). Front Mol Biosci 9, 931946. https://doi.org/10.3389/fmolb.2022.931946
Nagaraj YN, Burkina V, Okmane L, Blomqvist J, Rapoport A, Sandgren M, Pickova J, Sampels S, Passoth V (2022) Identification, quantification and kinetic study of carotenoids and lipids in Rhodotorula toruloides CBS14 cultivated on wheat straw hydrolysate. Fermentation 8, 300. https://doi.org/10.3390/fermentation8070300
Martín-Hernandez GC, Müller B, Brandt C, Hölzer M, Viehweger A, Passoth V (2022). Near chromosome-level genome assembly and annotation of Rhodotorula babjeveae strains reveals high intraspecific divergence. J Fungi 8, 323. https://doi.org/10.3390/jof8040323
Martín-Hernandez GC, Müller B, Chmielarz M, Brandt C, Hölzer M, Viehweger A, Passoth V (2021). Chromosome-level genome assembly and transcriptome-based annotation of the oleaginous yeast Rhodotorula toruloides CBS 14. Genomics 113, 4022-4026. https://doi.org/10.1016/j.ygeno.2021.10.006
Brandenburg J, Blomqvist J, Shapaval V, Kohler A, Sampels S, Sandgren M, Passoth V (2021) Oleaginous yeasts respond differently to carbon sources present in lignocellulose hydrolysate. Biotechnol Biofuels 14, 124. https://doi.org/10.1186/s13068-021-01974-2
Chmielarz M, Blomqvist J, Sampels S, Sandgren M, Passoth V (2021) Microbial lipid production from crude glycerol and hemicellulosic hydrolysate with oleaginous yeasts. Biotechnol Biofuels 14, 65. https://doi.org/10.1186/s13068-021-01916-y
Laurent J, Struyf N, Bautil A, Bakeeva A, Chmielarz M, Lylyy M, Herrera-Malaver B, Passoth V, Verstrepen KJ, Courtin CM (2021) The potential of Kluyveromyces marxianus to produce low-FODMAP straight-dough and sourdough bread: a pilot study. Food Bioprocess Technol 14, 1920-1935. https://doi.org/10.1007/s11947-021-02681-4
Sanya DRA, Onésime D, Passoth V, Maiti MK, Chattopadhyay A, Khot MB 2021. Yeast of the Blastobotrys genus are promising platform for lipid- based fuels and oleochemicals production. Appl Microbiol Biotechnol 105, 4879-4897. https://doi.org/10.1007/s00253-021-11354-3
Lapeña D, Kosa G, Hansen LD, Mydland LT, Passoth V, Horn SJ, Eijsink VGH (2020) Production and characterization of yeasts grown on media composed of spruce-derived sugars and protein hydrolysates from chicken by-products. Microbial Cell Factories 19. 19. https://doi.org/10.1186/s12934-020-1287-6
Lapeña D, Olsen PM, Arntzen MØ, Kosa G, Passoth V, Eijsink VGH, Horn SJ (2020). Spruce sugars and poultry hydrolysate as growth medium in repeated fed-batch fermentation processes for production of yeast biomass. Bioproc Biosyst Engin 43, 723-736. https://doi.org/10.1007/s00449-019-02271-x
Olsen M, Lindqvist R, Bakeeva A, Leong SL, Sulyok M (2019) Distribution of mycotoxins produced by Penicillium spp. inoculated in apple jam and crème fraiche during chilled storage. Int J Food Microb 292, 13-20. https://doi.org/10.1016/j.ijfoodmicro.2018.12.003
Chmielarz M, Sampels S, Blomqvist J, Brandenburg J, Wende F, Sandgren M, Passoth V (2019) FT-NIR: a tool for rapid intracellular lipid quantification in oleaginous yeasts. Biotechnol Biofuels 12:169. doi: 10.1186/s13068-019-1513-9. https://doi.org/10.1186/s13068-019-1513-9
Shapaval V, Brandenburg J, Blomqvist J, Tafintseva V, Passoth V, Sandgren M, Kohler A (2019). Biochemical profiling, prediction of total lipid content and fatty acid profile in oleaginous yeasts by FTIR spectroscopy. Biotechnol Biofuels 12, 140. doi: 10.1186/s13068-019-1481-0. https://doi.org/10.1186/s13068-019-1481-0
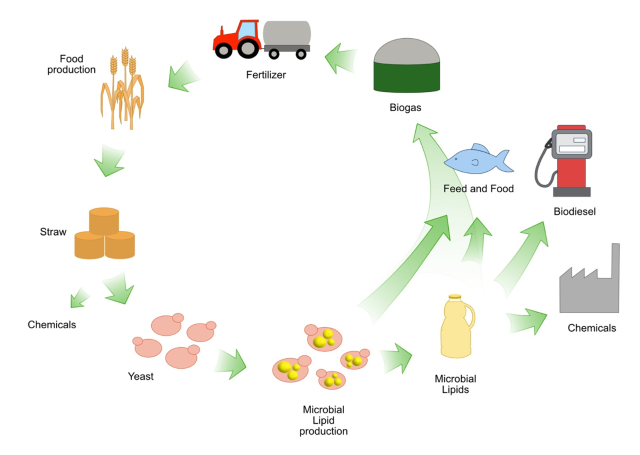
Passoth V, Sandgren M (2019). Biofuel production from straw hydrolysates: current achievements and perspectives. Appl Microbiol Biotechnol 103, 5105-5116. https://doi.org/10.1007/s00253-019-09863-3
Blomqvist J, Pickova J, Tilami SK, Sampels S, Mikkelsen N, Brandenburg J, Sandgren M, Passoth V (2018). Oleaginous yeast as a component in fish feed. Sci Rep. 8, 15945. https://www.nature.com/articles/s41598-018-34232-x
Brandenburg J, Poppele I, Blomqvist J, Puke M, Pickova J, Sandgren M, Rapoport A, Vedernikovs N, Passoth V (2018) Bioethanol and lipid production from the enzymatic hydrolysate of wheat straw after furfural extraction. Appl Microbiol Biotechnol 102, 6269-6277. https://doi.org/10.1007/s00253-018-9081-7
Karlsson H, Ahlgren S, Sandgren M, Passoth V, Wallberg O, Hansson PA (2017). Greenhouse gas performance of biochemical biodiesel production from straw- soil organic carbon changes and time-dependent climate impact. Biotechnol Biofuels 10, 273. https://doi.org/10.1186/s13068-017-0907-9
Karlsson H, Ahlgren S, Sandgren M, Passoth V, Wallberg O, Hansson PA (2016). A systems analysis of biodiesel production from wheat straw using oleaginous yeast: process design, mass and energy balances. Biotechnol Biofuels 9, 229. https://doi.org/10.1186/s13068-016-0640-9
Brandenburg J, Blomqvist J, Pickova J, Bonturi N, Sandgren M, Passoth V. (2016). Lipid production from hemicellulose with Lipomyces starkeyi in a pH regulated fed batch cultivation. Yeast 33, 451-462. doi: 10.1002/yea.3160. https://doi.org/10.1002/yea.3160