Contact
volkmar.passoth@slu.se, 018-673380
Non-conventional yeasts (i.e. all known yeast species except Saccharomyces cerevisiae, Candida albicans and Schizosaccharomyces pombe) are a currently under-utilised resource with a great academic and industrial potential.
The non-conventional yeast Dekkera bruxellensis outcompeted the traditional ethanol production yeast S. cerevisiae in glucose limited, high cell density industrial and laboratory scale ethanol fermentations, although it is growing slower than S. cerevisiae. This competitiveness is probably due to a more efficient metabolism and/ or more efficient transport. D. bruxellensis had a similar tolerance to and ability to ferment lignocellulose hydrolysate compared to industrial S. cerevisiae strains.
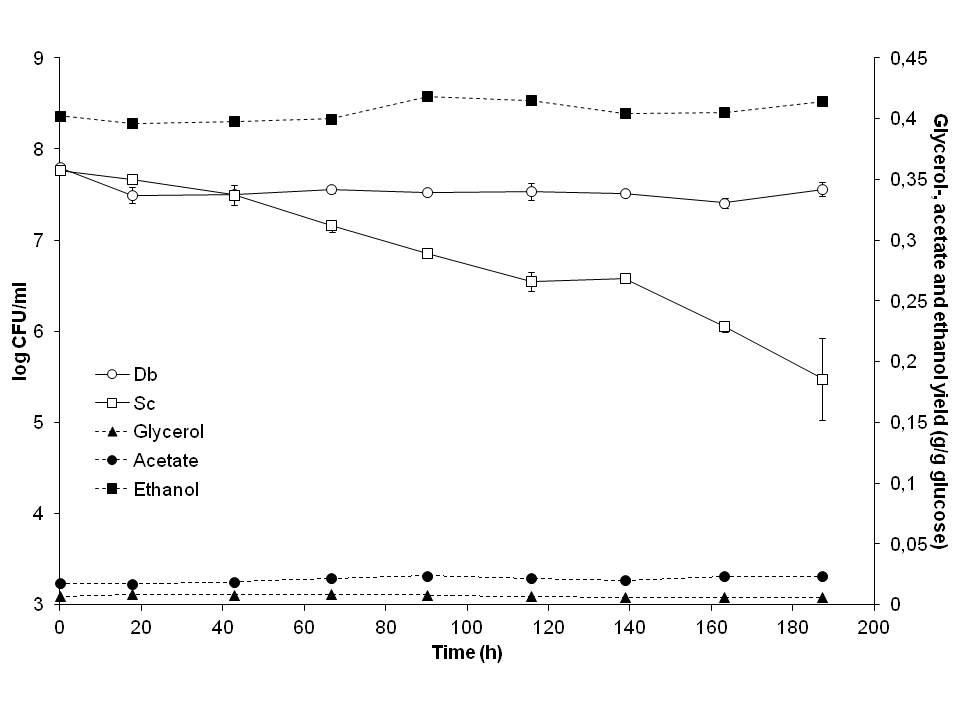
Co-culture of D. bruxellensis (Db) and S. cerevisiae (Sc) in continuous, glucose limited fermentation (modified from Blomqvist et al. 2012).
We are investigating cellular responses to substrate limitation, stressful substrates (lignocellulose) and bacterial contaminations on the transcriptional level, using next generation sequencing technology, enzyme activity determination and fermentation techniques. Specific metabolic and regulatory pathways for sugar uptake, energy metabolism, detoxification mechanisms and response to bacterial contamination will be identified. This will result in a comprehensive understanding of evolution of different yeast strategies to cope with environmental stress and in applied terms to a theory based selection of strains and fermentation conditions in industrial processes instead of the currently used trial and error strategy.
We are also performing physiological investigation of the xylose fermenting yeast Scheffersomyces (Pichia) stipitis within international collaborations.
Olsen RA, Bunikis I, Tiukova I, Holmberg K, Lötstedt B, Vinnere-Pettersson O, Passoth V, Käller M, Vezzi F (2015). De novo assembly of Dekkera bruxellensis: a multi technology approach using short and long-read sequencing and optical mapping. Gigascience 4, 56
Blomqvist J, Passoth V (2015). Dekkera bruxellensis- spoilage yeast with biotechnological potential, and a model for yeast evolution, physiology and competitiveness. FEMS Yeast Res 15, fov021
Theuretzbacher F, Blomqvist J, Lizasoain J, Klietz J, Potthast A, Horn SJ, Nilsen PJ, Gronauer A, Passoth V, Bauer A (2015). The effect of a combined biological and thermo-chemical pretreatment of wheat straw on energy yields in coupled ethanol and methane generation. Bioresource Technol 194, 7-13
Tiukova I, Eberhard T, Passoth V (2014). Interaction of Lactobacillus vini with the ethanol producing yeasts Dekkera bruxellensis and Saccharomyces cerevisiae. Biotechnol Appl Biochem 61, 40-44
Tiukova I, de Barros Pita W, Sundell D, Momeni MH, Horn SJ, Ståhlberg J, de Morais Jr MA, Passoth, V (2014) Adaptation of Dekkera bruxellensis to lignocellulose-based substrate. Biotechnol Appl Biochem 61, 51-57
Passoth V (2014). Bioethanol and other biofuels. In J Piskur and C Compagno (eds): Molecular Mechanisms in Yeast Carbon Metabolism. Springer Heidelberg, Berlin. pp. 217-259
de Barros Pita W, Silva DC, Simões DA, Passoth V, de Morais Jr MA (2013). Physiology and gene expression profiles of Dekkera bruxellensis in response to carbon and nitrogen availability. Antonie van Leeuwenhoek 104, 855-868
Tiukova IA, Petterson ME, Tellgren-Roth C, Bunikis I, Eberhard T, Pettersson OV, Passoth V (2013). Transcriptome of the alternative ethanol production strain Dekkera bruxellensis CBS 11270 in sugar limited, low oxygen cultivation. PLoS One 8: e58455
de Barros Pita W, Tiukova I, Leite FC, Passoth V, Simões DA, de Morais MA Jr. (2013). The influence of nitrate on the physiology of the yeast Dekkera bruxellensis grown under oxygen limitation. Yeast 30: 111-117
Passoth V, Rizwan MR, Nair HA, Olstorpe M, Tiukova I, Ståhlberg J 2013. Enhanced ethanol production from wheat straw by integrated storage and pre-treatment (ISP). Enzyme Microb. Technol. 52: 105-110
Blomqvist J, Nogue VS, Gorwa-Grauslund M, Passoth V 2012. Physiological requirements for growth and competitiveness of Dekkera bruxellensis under oxygen-limited or anaerobic conditions. Yeast, 29: 265-274
Blomqvist J, South E, Tiukova I, Momeni MH, Hansson H, Ståhlberg J, Horn SJ, Schnürer J, Passoth V. 2011. Fermentation of lignocellulosic hydrolysate by the alternative industrial ethanol yeast Dekkera bruxellensis. Lett Appl Microbiol, 53: 73-78
Dererie DY, Trobro S, Momeni MH, Hansson H, Blomqvist J, Passoth V, Schnürer A, Sandgren M, Ståhlberg J. 2011. Improved bio-energy yields via sequential ethanol fermentation and biogas digestion of steam exploded oat straw. Bioresour Technol 102: 4449-4455
Freese S, Passoth V, Klinner U. 2011. A mutation in the COX5 gene of the yeast Scheffersomyces stipitis alters utilization of amino acids as carbon source, ethanol formation and activity of cyanide insensitive respiration. Yeast 28: 309-320
Freese S, Vogts T, Speer F, Schäfer B, Passoth V, Klinner U. 2011. C- and N-catabolic utilization of tricarboxylic acid cycle-related amino acids by Scheffersomyces stipitis and other yeasts. Yeast 28: 375-390
Blomqvist J, Eberhard T, Schnürer J, Passoth V. 2010. Fermentation characteristics of Dekkera bruxellensis strains. Appl Microbiol Biotechnol. 87, 1487-1497
Maassen N, Freese S, Schruff B, Passoth V, Klinner U. 2008. Nonhomologous end joining and homologous recombination DNA repair pathways in integration mutagenesis in the xylose-fermenting yeast Pichia stipitis. FEMS Yeast Res. 8, 735-743
Jeffries TW, Grigoriev I, Grimwood J, Laplaza JM, Aerts A, Salamov A, Schmutz J, Lindquist E, Dehal P, Shapiro H, Jin YS, Passoth V, Richardson PM. 2007. Genome sequence of the lignocellulose-bioconverting and xylose-fermenting yeast Pichia stipitis. Nature Biotech. 25, 319-326
Passoth V, Blomqvist J, Schnürer J. 2007. Dekkera bruxellensis and Lactobacillus vini form a stable ethanol-producing consortium in a commercial alcohol production process. Appl Environ Microbiol. 73, 4354-4356
Görgens JF, Passoth V, van Zyl WH, Knoetze JH, Hahn-Hägerdal B. 2005. Amino acid supplementation, controlled oxygen limitation and sequential double induction improve heterologous xylanase production by Pichia stipitis. FEMS Yeast Res. 5, 677-683
Klinner U, Fluthgraf S, Freese S, Passoth V. 2005. Aerobic induction of respiro-fermentative growth by decreasing oxygen tensions in the respiratory yeast Pichia stipitis. Appl Microbiol Biotechnol 67, 247-253
Passoth V, Cohn M, Schäfer B, Hahn-Hägerdal B, Klinner U. 2003. Analysis of the hypoxia-induced ADH2 promoter of the respiratory yeast Pichia stipitis reveals a new mechanism for sensing of oxygen limitation in yeast. Yeast 20: 39-51
volkmar.passoth@slu.se, 018-673380
Funded by the Swedish Energy Authority (STEM) and the MicroDrivE-programme.