Experiments 2022
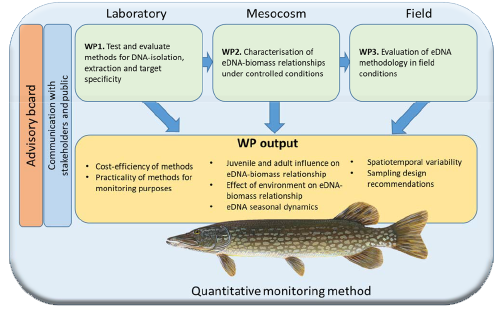
The large fieldworks within ePIKE are now completed, but we are still very active. During 2022 we have spent a lot of time in the lab, working on the development of a qPCR-assay targeting a multi-copy nuclear genetic marker instead of the more conventional mitochondrial markers. We hope that this approach will allow for equal, or even higher sensitivity making it possible to quantify considerably lower eDNA concentrations and provide a viable alternative to mitochondrial markers. Moreover, in combination with a mitochondrial marker we also anticipate to be able to detect active spawning events by studying the change in ratio between nuclear and mitochondrial gene copy numbers.
Results during 2022
We are very happy and proud to have the first ePIKE-publication coming out. Based on results from our earlier experiments it shows that the methods we have developed can quantify pike biomass, at least under controlled laboratory conditions (Karlsson et al. 2022).
We have also published a preprint of the fieldwork from 2020, where we collaborated with the ReFisk-project and compared pike abundance in spawning areas during spring (Ogonowski et al. 2022). Rod-fishing (i.e. angling) and eDNA showed a positive correlation, indicating that both methods can separate low-high pike abundance also in the field. However, we found that water temperature was very important for this relationship. Since there was more fish spawning with increasing temperature, thus releasing more DNA in to the water and increasing pike movements within the bays, we found an increased eDNA signal with increasing temperature. That rod-fishing and eDNA give slightly different results is not surprising since eDNA has the potential to sample a larger part of the population than angling, which is restricted to larger fish that are willing to strike the lure. The study also highlighted at what spatio-temporal scales variation in the eDNA signal was largest, which provides useful information for our continued work on developing a useful monitoring tool for Northern pike.
Ongoing 2023
The modified qPCR-assay using a nuclear genetic marker is currently being evaluated and optimized. Preliminary results indicate that the assay is, in all practical terms species specific and approximately 16 times more sensitive compared to the mitochondrial COI marker. The results of this study are expected to be published later this year.
The time-series data collected during 2021 from Hemmesta träsk combined with census data from the fish counter are currently being analysed with results expected later this year. We look forward to these results with excitement.
Publications
Karlsson, E., Ogonowski, M., Sundblad, G., Sundin, J., Svensson, O., Nousiainen, I., Vasemägi, A., 2022. Strong positive relationships between eDNA concentrations and biomass in juvenile and adult pike (Esox lucius) under controlled conditions: Implications for monitoring. Environmental DNA 4, 881–893. https://doi.org/10.1002/edn3.298
Ogonowski, M., Karlsson, E., Vasemägi, A., Sundin, J., Bohman, P., Sundblad, G., 2022. Temperature moderates eDNA-biomass relationships in northern pike. https://doi.org/10.1101/2022.12.28.522080